Gut health: GUT-MICROBIOTA-BRAIN connection
The Gut-Brain Connection: How Gut Health Influences Your Mind and Emotional Well-being
The connection between our gut and brain is something we all have felt. For example: think about the butterflies in your stomach when you’re excited, or the nausea and loss of appetite that can accompany intense anxiety. These are examples of how our emotions directly affect our gut, demonstrating the close relationship between our digestive system and the brain. In fact, our gut is not only sensitive to emotions but also to our thoughts and actions. Certain types of therapy, like cognitive-behavioral therapy, emphasize how connected our thoughts, feelings, and actions are, which also impacts gut health.
However, not only does our mental state affect our gut, but the reverse also happens: gut health influences our brain. To start, let’s consider a couple of fascinating facts:
- Our gut contains around 200 million neurons.
- Approximately 90-95% of the body’s serotonin (a key neurotransmitter that influences mood) is produced in the gut.
How the Gut and Brain Communicate: Key Players in the Gut-Brain Axis [1]

Photo: The Microbiota-Gut-Brain Axis (2019) [1]
The gut-brain axis is mainly composed of:
- Central Nervous System (CNS)
- Autonomic Nervous System (ANS)
- Enteric Nervous System (ENS)
- Hypothalamic-Pituitary-Adrenal (HPA) Axis
Communication between the brain and the gut occurs through the ANS, primarily via the vagus nerve, which is the longest autonomic nerve in our body. This nerve starts in the brainstem, passes through the neck, chest, and abdomen, and ends in the colon. In this way, it plays a critical role in regulating heart rate, blood pressure, digestion, and even speech.
This bidirectional communication system works through the release of neurohormones into the bloodstream and also involves the immune system, which communicates with the brain through signals like interleukin and interferon-gamma.
On the other hand, our gut microbiota, made up of billions of bacteria and other microorganisms in the gut, also influences both physical and mental health, demonstrating how crucial gut health is for overall well-being.
How It Works: Understanding the Enteric Nervous System and Gut Functions [1]
The enteric nervous system (ENS), also known as the “second brain”, is a complex network of neurons that extends from the esophagus to the anus. Therefore, it is a vital communication center, receiving sensory, sympathetic, and parasympathetic inputs that help regulate almost all functions in the gut. As a result, it receives information from the intestinal lining and sends it back to the brain.
Similarly, the vagus nerve plays a central role in managing gut function, including motility, mucus secretion, and blood flow. Here’s how the autonomic nervous system works in relation to the ENS:
- Parasympathetic Stimulation (via the vagus nerve): Increases gastrointestinal motility and secretion.
- Sympathetic Stimulation: Decreases gastrointestinal activity, balancing digestive function according to current needs.
Key Functions of the Gastrointestinal Tract
The gastrointestinal tract has two main functions:
- Digestion and Absorption: It is responsible for digesting and absorbing nutrients while managing the transport of water and electrolytes in the intestines.
- Defense: The gastrointestinal tract acts as a protective barrier, preventing harmful substances such as viruses, bacteria, and pro-inflammatory molecules from entering the bloodstream.
Defensive Barriers
From the outermost layers to the innermost, the protective barrier of the gut consists of:
- Microbiota
- Mucus
- Epithelial cells
- Immune cells
This selective barrier allows beneficial nutrients to pass while blocking harmful agents.

Photo: The Microbiota-Gut-Brain Axis (2019) [1]
Gut Immunity: The Role of GALT
The gut immune system, known as GALT (Gut-Associated Lymphoid Tissue), distinguishes between harmless substances and harmful invaders. When a threat is detected, it triggers a defense response, launching inflammatory reactions as needed. Therefore, the microbiota and mucus are essential parts of this immune barrier, protecting not only against infections but also preventing toxins from entering the bloodstream.
However, alterations in this system can lead to conditions like:
- Irritable Bowel Syndrome (IBS)
- Excessive candida growth
- Small Intestinal Bacterial Overgrowth (SIBO)
- Celiac disease
Additionally, these imbalances can trigger immune responses, increase intestinal permeability, and activate pain pathways in the ENS. As a result, these alterations can lead to dysregulation of the gut-brain axis, affecting motility, secretion, and causing visceral hypersensitivity.
Gut Inflammation Impacts Brain Health
Every time we eat, a mild inflammatory response is triggered as a precautionary measure, which typically resolves after digestion. On the other hand, chronic inflammation due to frequent eating or consuming certain foods can create an inflammatory environment. In this case, when inflammation becomes chronic, stress signals can be sent to the brain, leading to symptoms such as:
- Fatigue and low energy
- Apathy and even depressive symptoms
Microbiota and Gut Health: A Key Player in Our Gut-Brain Connection
Our microbiota consists of approximately 10 trillion microorganisms, including bacteria, protozoa, fungi, yeasts, and parasites. While it was previously believed that 90% of our cells were bacterial, new evidence suggests a near 50-50 ratio with human cells. [2]
This means that for every human cell, there is approximately one bacterial cell.
The Role of Microbiota in Brain Development and Function
In terms of the role of microbiota in brain development and function, studies show that bacterial colonization in the gut is crucial for the development and maturation of both the enteric nervous system (ENS) and the central nervous system (CNS). However, the absence of this colonization is linked to alterations in neurotransmitter expression, leading to delays in gastric emptying and intestinal transit. [3]
Similarly, research also links gut dysbiosis (imbalance in microbial populations) with mental health disorders such as:
- Depression
- Anxiety
- Neurodegenerative conditions like Alzheimer’s and Parkinson’s
Furthermore, dysbiosis can influence gastrointestinal motility, secretion, and even visceral sensitivity, which can have significant effects on behavior and emotional health. [4], [5], [6], [7]
On the other hand, the microbiota communicates with the brain through various mechanisms, including the release of molecules into the bloodstream, activation of immune cells, and direct stimulation of the vagus nerve. This influences the release of neurotransmitters, which in turn can affect mental health.
Microbiota, Gut Health, and Human Behavior
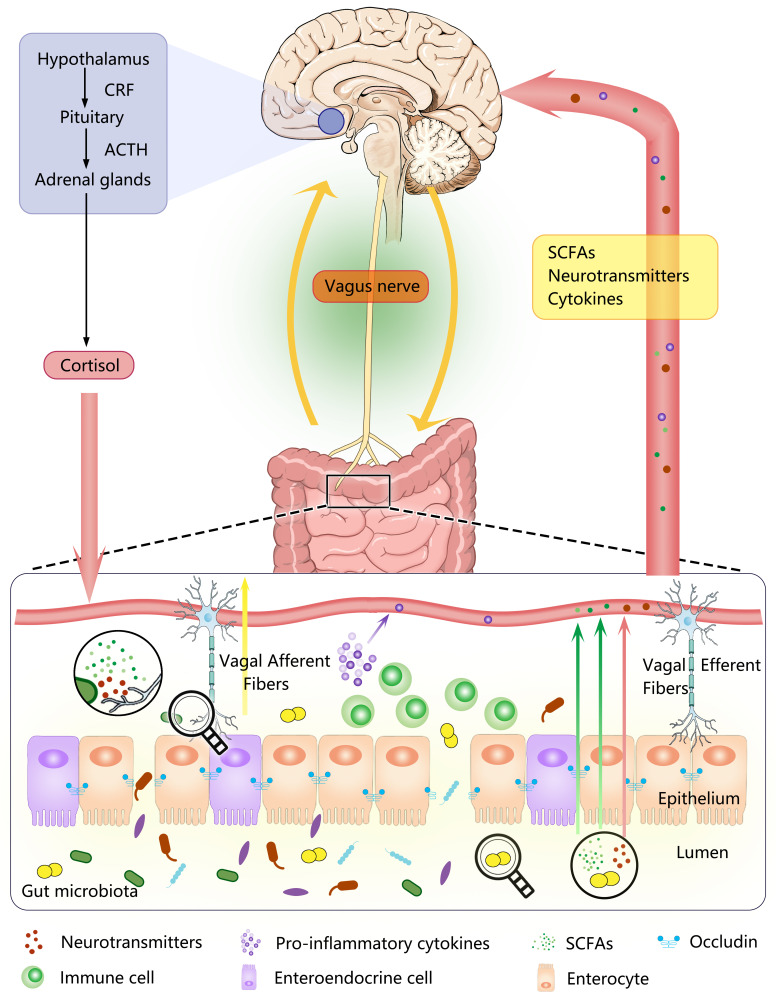
Photo: Vagus Nerve and Underlying Impact on the Gut Microbiota-Brain Axis in Behavior and Neurodegenerative Diseases (2022) [8]
Changes in the microbiota can affect mood and behavior, and conversely, behavior changes or stress can alter the composition of the microbiota. Communication between the microbiota and the brain occurs through multiple pathways:
The Vagus Nerve
This nerve acts as a bridge between the gut and the brain, detecting changes in the microbiota and transmitting them to the brain. Additionally, certain pathogens and proteins can even travel along the vagus nerve. Research has shown that vagus nerve stimulation can help treat conditions like depression, chronic pain, anxiety, and stress.
For example, studies that administered Lactobacillus rhamnosus to mice showed an increase in the transcription of γ-aminobutyric acid (GABA), which changed behavior based on vagal integrity. [8]
Neurohormones
Neurohormones like serotonin, dopamine, and catecholamines, which are largely produced in the gut, also play a role in mood and behavior.
For instance, 90% of the body’s serotonin, which influences mood, is produced in the gut and is regulated by the microbiota. Although serotonin produced in the gut does not directly cross the blood-brain barrier (BBB), it impacts mood by regulating precursors that do, such as tryptophan, whose production is regulated by enzymes metabolized by the gut microbiota.
Moreover, dopamine production is also linked to the microbiota. Studies in germ-free mice have shown reduced dopamine levels, which is an interesting area of research for understanding the pathogenesis of Parkinson’s disease. [4], [9]
The microbiota contributes to the production of GABA. In this regard, certain bacteria that contain the enzyme glutamate decarboxylase convert dietary glutamate into GABA, an essential neurotransmitter for behavior modulation. Studies indicate that supplementation with probiotics, which increases GABA availability, may improve anxiety control, highlighting the role of the microbiota in mental health. [10]
The Impact of Short-Chain Fatty Acids (SCFAs) on Brain Function
- Short-chain fatty acids (SCFAs), produced through the digestion of dietary fibers by the microbiota, are capable of crossing the BBB and reaching the hypothalamus, influencing neurotransmitters like GABA and glutamate. [11]
- The hypothalamic-pituitary-adrenal (HPA) axis: regulates our response to stress. Studies conducted with germ-free mice have observed exaggerated inflammatory responses to stress, suggesting that a healthy microbiota can help moderate these responses. For example, high levels of Lactobacillus rhamnosus have been associated with lower levels of corticosterone (a stress hormone), better stress management, reduced depression, and decreased inflammation. Conversely, small exposures to stress can affect the microbiota, showing how deeply interconnected these systems are. [11]
- Release of anti-inflammatory cytokines, which can cross the blood-brain barrier (BBB) and stimulate neurogenesis. It is believed that this process is neuroprotective, especially after events such as ischemic injury.
When Dysbiosis Hits
When dysbiosis occurs, the concentrations of important molecules can be altered. This, in turn, can contribute to the development of various diseases:
-
Behavioral disorders and anxiety
-
Alzheimer’s, Parkinson’s
-
Schizophrenia and autism
Therefore, understanding and maintaining a healthy microbiota is not only crucial for gut health, but it can also be key to supporting emotional and cognitive well-being. [4], [12], [13], [14]
Emotional Behavior and Obesity: The Role of Gut Microbiota
In overweight and obese individuals, studies reveal a dysbiotic gut profile, often with reduced microbial diversity. In fact, dysbiosis related to obesity leads to metabolic complications, affecting immunity, energy regulation, intestinal hormones, and inflammatory processes. [15]
We know that short-chain fatty acids (SCFAs) produced by intestinal bacteria, such as butyrate, promote the health of colon cells, while propionate regulates satiety through liver functions, and acetate influences cholesterol metabolism and appetite regulation. [16]

Photo: The Microbiota-Gut-Brain Axis (2019) [1]
Stress and Anxiety: The Impact on the Gut-Brain Axis
Prolonged exposure to stress can lead to overactivation of the paraventricular nucleus in the hypothalamus, resulting in elevated cortisol levels in the blood. This increase in cortisol can alter key brain regions such as the hippocampus, amygdala, and prefrontal cortex, affecting the HPA axis’s ability to regulate stress. In fact, inadequate inhibition of the HPA leads to high and sustained cortisol levels, contributing to various conditions, including Irritable Bowel Syndrome (IBS). Studies indicate that individuals with diarrhea-predominant IBS show greater cortisol reactivity compared to those with constipation-predominant IBS or healthy controls. [17], [18]
Furthermore, stress also affects gastrointestinal functions, influencing permeability, motility, visceral sensitivity, blood flow, secretion, and microbiota composition. Underlying mechanisms to these alterations include cortisol increases, activation of the sympathetic nervous system, dysfunction of the intestinal barrier, and stimulation of immune cells. Stress is associated with several gastrointestinal issues, such as:
-
Gastroesophageal reflux
-
Peptic ulcers
-
Inflammatory bowel disease
-
Food intolerances [1]
For example, acute stress events, like public speaking, are known to alter gastric acid and mucus secretions, which can increase intestinal permeability and discomfort.
Stress in Childhood and Its Long-Term Effect on Gut Health
Recent studies reveal that early childhood stress, such as maternal separation in neonates, can lead to increased serotonin production in the gut, correlating with IBS symptoms, especially in diarrhea-predominant cases. [19]
Regarding microbiota, it has been shown that maternal stress during pregnancy has been linked to dysbiosis in babies, characterized by an increase in Proteobacteria and a decrease in beneficial bacteria, generating:
-
Gastrointestinal symptoms
-
Increased risk of neurodevelopmental disorders
Additionally, galanin, a neuropeptide present in the gut and central nervous system, stimulates cortisol release and affects intestinal motility, pancreatic function, and growth hormone levels. Therefore, the intestinal microbiota can activate the mucosal immune system, a critical process in intestinal immune disorders.
In the context of the gut-brain-microbiota axis, the brain manages gut functions like motility, secretion, and immune responses. However, stress disrupts these functions, altering the quality and quantity of mucus, which affects the microbial habitat in the gut.
The gut microbiota also affects the production of gamma-aminobutyric acid (GABA).
GABA is a neurotransmitter that calms brain activity and alleviates anxiety. Due to its calming effects, GABA is commonly found in supplements aimed at managing anxiety and improving sleep quality.
Interestingly, research suggests that women who consume probiotics may exhibit reduced sensitivity to negative emotional stimuli, highlighting the gut-brain connection. Additionally, certain bacterial strains, such as Campylobacter jejuni, have been linked to an increase in anxiety, illustrating how imbalances in the microbiota can affect mental health. [20]
The Role of Melatonin in Gut Health and Stress [21]
Melatonin, known primarily for regulating sleep, also plays a crucial role in gastrointestinal health. Although it is synthesized and released by the pineal gland into the bloodstream, reaching its peak secretion at night, melatonin levels in the gastrointestinal tract are approximately 400 times higher than in the pineal gland.
Research indicates that melatonin helps prevent and heal stress-induced ulcers by acting directly on the gut. For example, a study showed that melatonin in the duodenum stimulates bicarbonate release to neutralize gastric acid entering from the stomach, a key response to stress-induced acidity.
Additionally, melatonin reduces inflammation in the gut by neutralizing reactive oxygen species and inducing antioxidant enzymes such as superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-PX). [22], [23]
The protective effects of melatonin against stress-induced gastrointestinal injuries are remarkable. In fact, it acts as an antioxidant, reduces inflammation, and modulates intestinal motility. In humans, an 8-week oral melatonin treatment has shown to relieve abdominal pain, bloating, and defecation problems in people with Irritable Bowel Syndrome (IBS). [24], [25]
Irritable Bowel Syndrome (IBS) and Its Multifactorial Etiology
IBS is a prevalent functional gastrointestinal disorder, affecting 10-20% of the global population. It is often associated with stress and linked to environmental factors, psychosocial stress, intestinal inflammation, and microbiota imbalances. IBS patients typically show higher sympathetic activity and reduced parasympathetic function, which affects motility and intestinal secretion. In cases of severe stress, vagus nerve dysregulation can even lead to gastrointestinal bleeding and peptic ulcers.
On the other hand, in patients with diarrhea-predominant IBS, certain probiotic strains have been shown to reduce intestinal permeability, highlighting the therapeutic potential of microbiota modulation. [17], [18]
Gut Health and Other Mental Disorders: Alzheimer’s, Depression, and Autism
In Alzheimer’s patients, supplementation with probiotics has shown promising results in improving cognitive function. [26], [27]
Dopamine is crucial for regulating motivation, pleasure, relaxation, and memory duration. Studies with germ-free mice have shown these animals have lower dopamine levels, indicating the influence of intestinal microbiota on its production.
Additionally, studies comparing the gut microbiota of 37 people with depression to 18 healthy controls revealed distinct microbial patterns. Depressed people had lower abundance of Bacteroidetes and higher presence of Alistipes, suggesting a possible link between the intestinal microbial composition and mood disorders. [28], [29]
Regarding autism, studies suggest a distinct microbiota profile in affected children, with notable differences compared to neurotypical peers. This line of research has the potential to open new therapeutic avenues for managing autism and related symptoms. [30], [31]
Conclusions
The concept of isolated systems within the human body is increasingly outdated. We’ve explored how truly interconnected the body’s systems are and how a holistic perspective improves our understanding of health and well-being. Viewing the body as an integrated whole, rather than a mere collection of separated parts, allows for a deeper understanding of its complex functions and interdependencies.
There are other fascinating “axes” within the body that significantly impact mental health, and exploring them could shed even more light on holistic wellness approaches.
If you found this article insightful and are curious about specific ways to diagnose, treat, and improve this axis, let me know in the comments; I’d be happy to delve into practical strategies in a future article. If you’d like to schedule a consultation, feel free to contact me by clicking here.
You would like to learn more? Here’s a podcast I took part in about how mental health and the gut are connected.
REFERENCES
- The Microbiota-Gut-Brain Axis (2019) https://journals.physiology.org/doi/full/10.1152/physrev.00018.2018?rfr_dat=cr_pub++0pubmed&url_ver=Z39.88-2003&rfr_id=ori%3Arid%3Acrossref.org](https://journals.physiology.org/doi/full/10.1152/physrev.00018.2018?rfr_dat=cr_pub++0pubmed&url_ver=Z39.88-2003&rfr_id=ori%3Arid%3Acrossref.org
- Revised Estimates for the Number of Human and Bacteria Cells in the Body (2016)
https://pmc.ncbi.nlm.nih.gov/articles/PMC4991899/ - Microbiota and neurodevelopmental windows: implications for brain disorders (2014)
https://pubmed.ncbi.nlm.nih.gov/24956966/ - Gut and Brain: Investigating Physiological and Pathological Interactions Between Microbiota and Brain to Gain New Therapeutic Avenues for Brain Diseases (2021)
https://pmc.ncbi.nlm.nih.gov/articles/PMC8545893/ -
Role of the gut microbiota in nutrition and health (2018)
- The gut-brain axis: interactions between enteric microbiota, central and enteric nervous systems (2015)
https://pubmed.ncbi.nlm.nih.gov/25830558/ - The Human Intestinal Microbiome in Health and Disease (2016)
https://www.nejm.org/doi/full/10.1056/NEJMra1600266 - Vagus Nerve and Underlying Impact on the Gut Microbiota-Brain Axis in Behavior and Neurodegenerative Diseases (2022)
https://pmc.ncbi.nlm.nih.gov/articles/PMC9656367/ - Changes of Dopamine and Tyrosine Hydroxylase Levels in the Brain of Germ-free Mice (2023)
https://www.ncbi.nlm.nih.gov/pmc/articles/pmid/36811101/ - Probiotic, Prebiotic, and Brain Development (2018)
https://pubmed.ncbi.nlm.nih.gov/29135961/ - Specialized metabolites from the microbiome in health and disease (SCFA) (2014)
https://pubmed.ncbi.nlm.nih.gov/25440054/ - The Gut-Brain Axis: Influence of Microbiota on Mood and Mental Health (2019)
https://pmc.ncbi.nlm.nih.gov/articles/PMC6469458/ - Intestinal microbiota and mental behavior disorders (2020)
http://scielo.sld.cu/scielo.php?script=sci_arttext&pid=S0034-75312020000200016 - Cognitive-Behavioural Correlates of Dysbiosis: A Review (2020)
https://pmc.ncbi.nlm.nih.gov/articles/PMC7402132/ -
Dysbiotic Gut Bacteria in Obesity: An Overview of the Metabolic Mechanisms and Therapeutic Perspectives of Next-Generation Probiotics (2022)
- The role of short chain fatty acids in appetite regulation and energy homeostasis (2015)
https://pmc.ncbi.nlm.nih.gov/articles/PMC4564526/ - Pathophysiology underlying irritable bowel syndrome -From the viewpoint of dysfunction of autonomic nervous system activity (2014)
https://www.jstage.jst.go.jp/article/jsmr/45/1/45_1_15/_pdf/-char/en - Irritable bowel syndrome: A microbiome-gut-brain axis disorder? (2014)
https://pmc.ncbi.nlm.nih.gov/articles/PMC4202342/ - The microbiome-gut-brain axis during early life regulates the hippocampal serotonergic system in a sex-dependent manner (2012)
https://www.nature.com/articles/mp201277 - Consumption of Fermented Milk Product With Probiotic Modulates Brain Activity (2013)
https://www.gastrojournal.org/article/s0016-5085(13)00292-8/fulltext - Distribution, function and physiological role of melatonin in the lower gut (2011)
https://pmc.ncbi.nlm.nih.gov/articles/PMC3198018/ - Melatonin in the duodenal lumen is a potent stimulant of mucosal bicarbonate secretion (2002)
https://www.researchgate.net/publication/10834201_Melatonin_in_the_duodenal_lumen_is_a_potent_stimulant_of_mucosal_bicarbonate_secretion - Antioxidant Actions of Melatonin: A Systematic Review of Animal Studies (2023)
https://pmc.ncbi.nlm.nih.gov/articles/PMC11047453/ - Melatonin improves abdominal pain in irritable bowel syndrome patients who have sleep disturbances: a randomised, double-blind, placebo-controlled study (2005)
https://pmc.ncbi.nlm.nih.gov/articles/PMC1774717/ - Melatonin improves bowel symptoms in female patients with irritable bowel syndrome: a double-blind, placebo-controlled study (2005)
https://onlinelibrary.wiley.com/doi/10.1111/j.1365-2036.2005.02673.x - Probiotics for Alzheimer’s Disease: A Systematic Review(2022)
https://pmc.ncbi.nlm.nih.gov/articles/PMC8746506/ - Effect of Probiotic Supplementation on Cognitive Function and Metabolic Status in Alzheimer’s Disease: A Randomized, Double-Blind and Controlled Trial (2016)
https://www.frontiersin.org/journals/aging-neuroscience/articles/10.3389/fnagi.2016.00256/full - Altered Composition of Gut Microbiota in Depression: A Systematic Review (2020)
https://pmc.ncbi.nlm.nih.gov/articles/PMC7299157/#B25 - Correlation between the human fecal microbiota and depression (2015)
https://pubmed.ncbi.nlm.nih.gov/24888394/
- Multi-level analysis of the gut–brain axis shows autism spectrum disorder-associated molecular and microbial profiles (2023)
https://pmc.ncbi.nlm.nih.gov/articles/PMC10322709/ - Global prevalence of autism spectrum disorder and its gastrointestinal symptoms: A systematic review and meta-analysis (2022)
https://pmc.ncbi.nlm.nih.gov/articles/PMC9445193/



Leave a Reply
Want to join the discussion?Feel free to contribute!